Next-gen therapeutics
The future of microbiome medicine
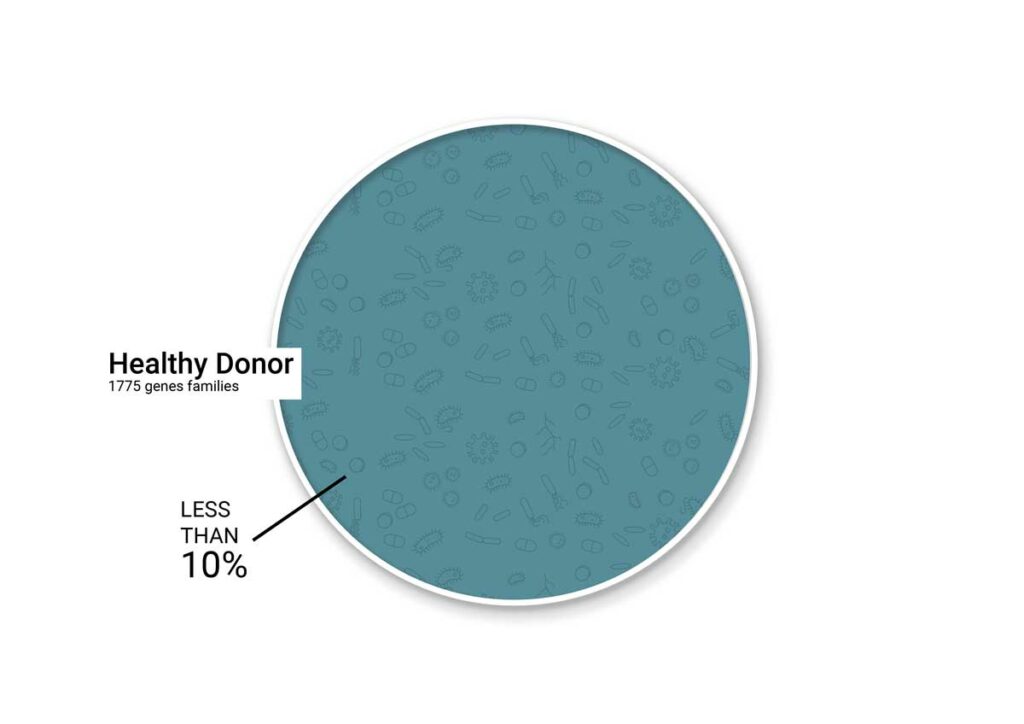
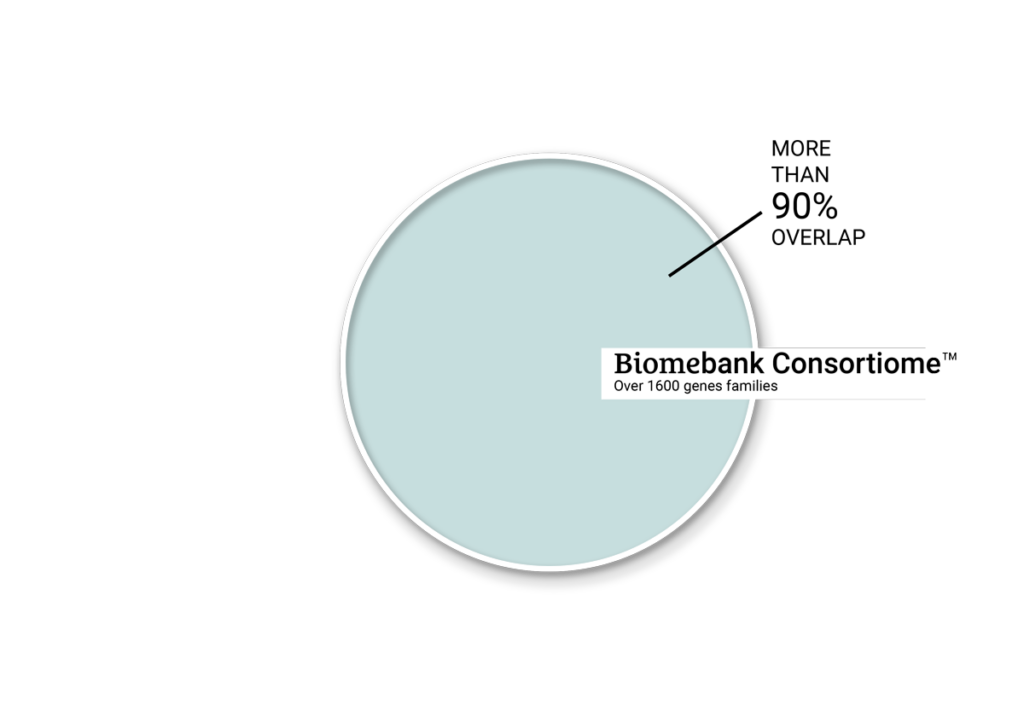
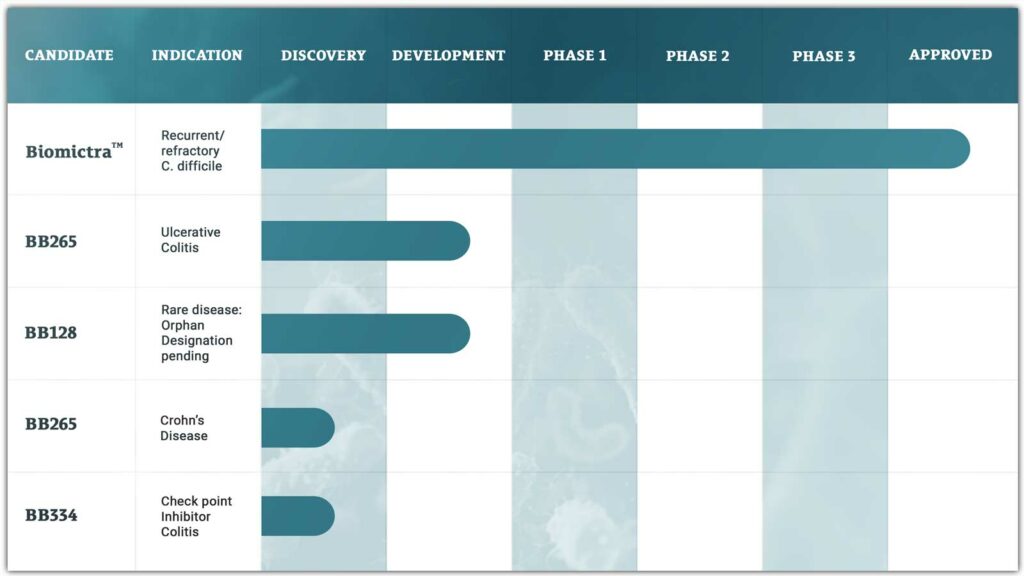
CONSORTIOME® is BiomeBank’s proprietary platform for next-generation microbial therapeutics. It builds complete, co-cultured gut communities that mirror the human microbiome, offering a scalable, precisely controlled alternative to donor-derived therapies. Guided by our bank of real-world clinical feedback, CONSORTIOME® delivers reproducible, high-quality interventions at scale, unlocking the next frontier in microbiome medicine.