This article was originally posted on Medical Daily here
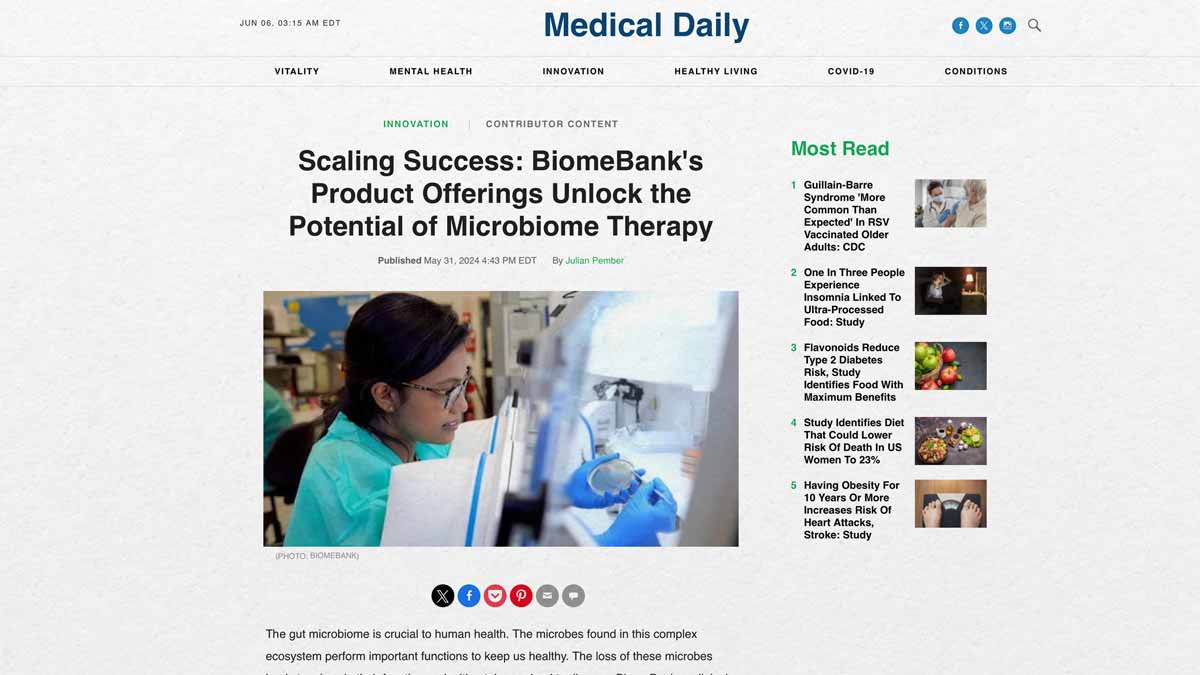
The gut microbiome is crucial to human health. The microbes found in this complex ecosystem perform important functions to keep us healthy. The loss of these microbes leads to a loss in their function and, ultimately, can lead to disease. BiomeBank, a clinical-stage biotechnology company, is on a mission to treat and prevent disease by restoring the gut microbial ecology.
BiomeBank’s donor-derived microbiome therapy, provides an alternative way of treating conditions like C. difficile infection than traditional treatment. With its approval by Australian regulators in 2022, BiomeBank became the first company to gain regulatory approval for a donor-derived microbiome drug product in the world.
BiomeBank’s drug product is a donor-derived microbiome therapy in which gut bacteria and other microbes of a healthy person are transplanted into another, helping restore the functionality of a healthy microbiome.
“BiomeBank’s donor-derived product is approved in Australia for the treatment of C. Difficile infection. However, we see the product’s greatest value as a tool to develop cultured microbiome therapies targeting specific diseases. We are using it in clinical trials where we can correlate positive clinical responses with microbiome data to identify organisms that carry a mechanism of action associated with treating the disease. We pull out those microbes from our large gut bacterial culture collection, and from those develop a cultured version of the therapy.” says CEO Sam Costello.
Equipped with the abundance of data and human gut microbes at their disposal, the team at BiomeBank created Consortiome™, a rationally designed microbial platform in which they are able to generate an artificial human gut microbial community in a bioreactor. It contains more than 90% of the known gene families found in a healthy person’s microbiome, providing the ability to deliver highly targeted disease-specific functions. Consortiome™ eliminates the reliance on stool donors, enabling BiomeBank to produce microbial therapies with diverse functional capability at scale, to large markets, and at a much lower cost than other microbial products.
Costello explains, “We have discovered that therapies developed with our Consortiome platform to contain a diverse composition of microbes, carry far more powerful functions than single microbes or small groups of microbes. We expect that these emergent functions will translate to improved efficacy of these microbiome therapies.”
The first cultured therapy is BB265 for the treatment of ulcerative colitis (UC). UC is a chronic disease in which the lining of the colon becomes inflamed and develops tiny open sores or ulcers. This results in symptoms like abdominal pain, diarrhea (sometimes bloody), and a feeling of urgency to have a bowel movement. In more severe cases, patients must undergo surgery to remove part of the bowel.
Because some of the treatments often prescribed for UC are immunosuppressive, they can lead to other complications, such as a higher risk of infections. These challenges with existing treatments led BiomeBank to find a better solution for the nearly one million people living with UC in the US alone.
When discussing BB265, Costello states, “We have conducted studies using Biomictra™ in patients with ulcerative colitis and showed that you could induce remission using that therapy.”
One common mechanism of action was identified among the patients who responded to the treatment. Using their culture collection, the BiomeBank team picked out the organisms with the identified mechanism and put together a cultured version that mimicked the donor composition and had the full spectrum of the gut microbe ecosystem, resulting in a therapy that is an enhanced version of the donor product
While the potential for BB265 is showing enormous promise for UC patients, the company is also using the same methodology to develop BB128, a therapy in an orphan disease indication. They are currently conducting a discovery trial using Biomictra™ to gather more data to formulate a disease specific therapy.
This article was originally posted on Medical Daily here