Page Contents
Overview
The more that I've been involved in research in the area, as well as clinical trials and treating patients, the more I've learned about the potential… and also the gravity of the problem associated with loss of gut microbes.
- Dr Samuel Costello, Chief Medical Officer
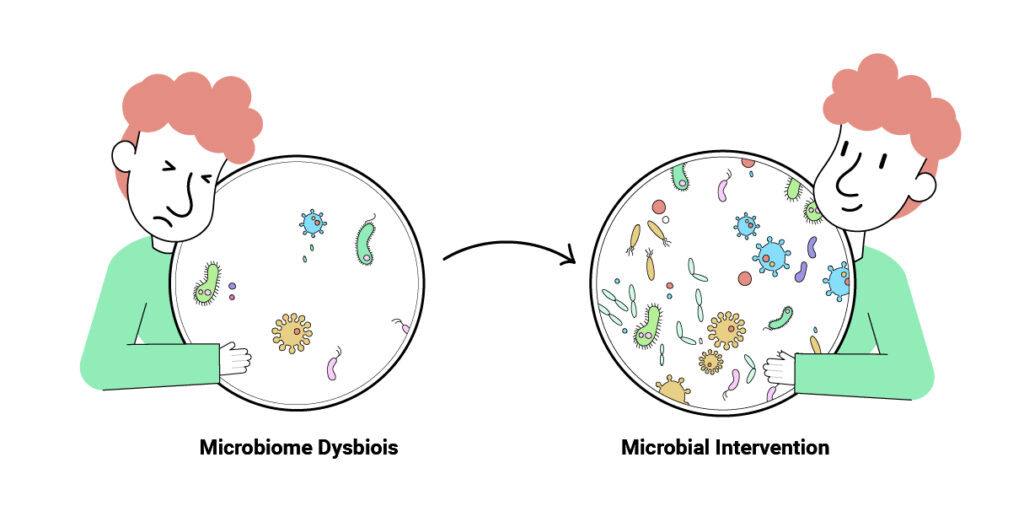
A Thriving Ecosystem
Treatments of the Future

The priority for microbiome therapeutics companies is learning how different microbial species influence our health. The goal is to develop targeted therapies that restore lost microbial function to treat microbiome-mediated chronic disease.
First Generation Treatments
BiomeBank is pioneering microbiome-based therapies which restore lost gut microbial ecology to treat and prevent chronic disease to help patients with unmet medical need.
For example, studies have demonstrated feacal microbiota transplantation (FMT) can effectively treat clostridioides difficile (C.difficile), a life threatening infection with cure rates of 81% – 96%. This procedure involves transplanting the gut microbial ecology from a healthy donor into the gut of a patient with chronic disease.
Microbiome-based therapies are currently being studied as a treatment for other inflammatory diseases such as Ulcerative Colitis. In a randomised clinical trial published in JAMA, BiomeBank’s Chief Medical Officer Dr Samuel Costello and his team demonstrated that FMT can induce clinical remission in patients with mild to moderate Ulcerative Colitis. A recent study in Lancet further demonstrated the role of microbiome-based therapies in treating Ulcerative Colitis (Haifer et al. 2022 Lancet).
The microbial ecology used to manufacture microbiome-based therapies can only be harvested from healthy human stool donors who have undergone a rigorous quality controlled screening and donation process. Microbiome-based therapies such as FMT are generally performed using colonoscopy. However, BiomeBank is provide a microbiome-based therapy in capsules. Promising research demonstrates the efficacy of delivering FMT via freeze-dried, oral capsules, which is significantly more cost-effective, less time-consuming and easier to scale-up for widespread clinical use.

WATCH BiomeBank’s Chief Medical Officer Dr Samuel Costello talk about his experiences of using Microbial Therapies with patients.
Second Generation Treatments
BiomeBank is developing second generation microbiome-based therapies that replicate the bacterial complexity seen in FMT while also containing target bacterial strains which carry important function for treating disease. This next generation microbiome-based therapy will be manufactured in bioreactors, freeze-dried, and encapsulated for global distribution. This is made possible by BiomeBank’s Consortiome Technology.
BiomeBank is currently developing second generation microbiome-based therapies for the treatment of recurrent C.difficile infection and ulcerative colitis.